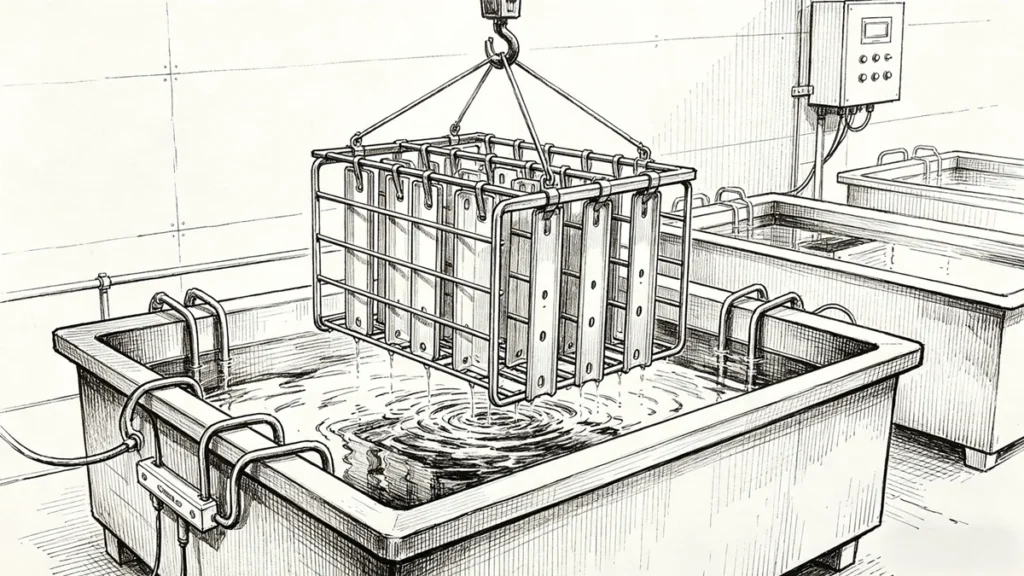
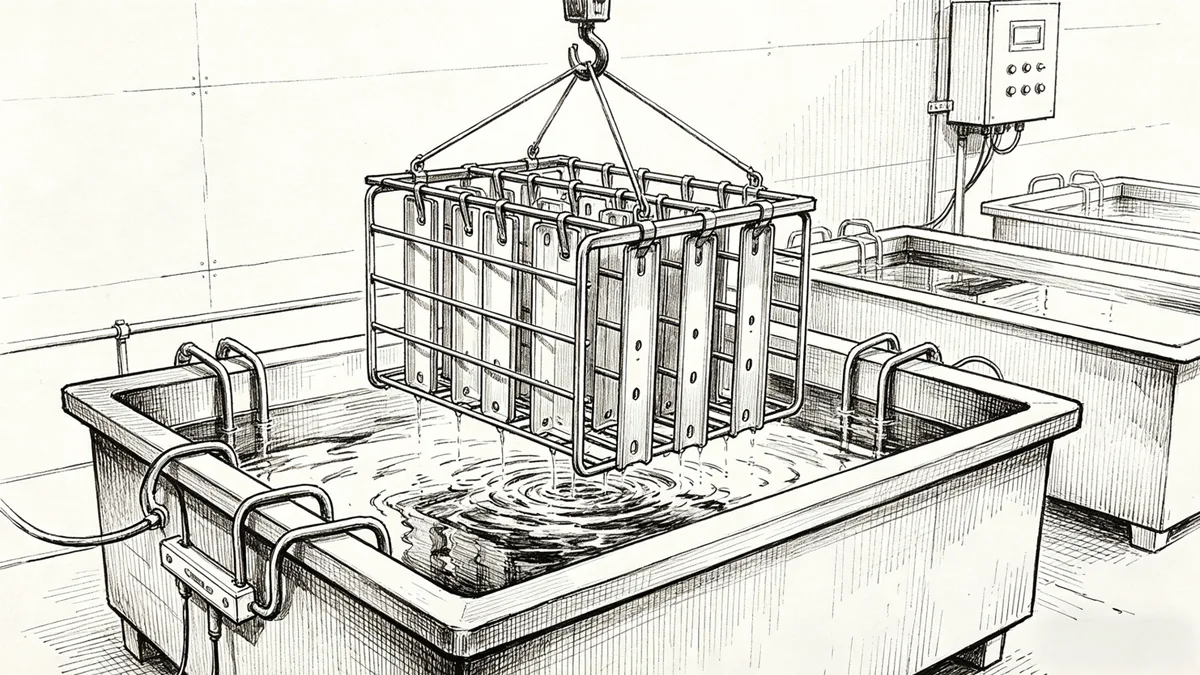
Anodizing is an electrochemical passivation that is employed to thicken the natural oxide coating on a metal’s surface, predominantly aluminum. In aluminum anodizing, the metal acts as an anode in an electrolytic cell, which is often placed into an acid electrolyte, and an oxidation is driven by a controlled electric current. This produces a thick, adsorbed layer of oxide- mostly aluminum oxide – that is part of the substrate and not a coating.
The anodizing process improves various vital performance features. It enhances surface hardness and wear resistance, which makes it useful in molds and jigs that are subjected to repetitive mechanical contact. Anodized aluminum parts in injection-molded tooling will have better release properties and less galling. Anodized surfaces can be functionally engineered to be electrically insulated or to conduct conductively, which is needed in electronic housings and heat sinks. Anozation leads to corrosion resistance and cleanliness in the medical and food-contact settings. Aesthetically, the porous oxide layer is able to take in dyes and therefore has a large selection of enduring colors and finishes.
What Materials Can and Cannot Be Anodized?
The most commonly anodized materials are aluminum and its alloys, because they have a natural tendency toward forming oxides [1]. Typical alloys that react to anodization of aluminum include 6061, 6063, and 7075, with the end finish being different based on the alloying elements, including silicon or copper. Special anodizing of titanium and magnesium can create a special oxide film, either functional or decorative.
The inability of metals like steel, copper, and brass to be anodized in the traditional sense is because they do not develop stable, protective oxide layers in anodic environments. Although they may withstand other surface treatments, e.g., electroplating or passivation, the actual anodizing metal processes are mostly limited to aluminum, titanium, magnesium, and some other reactive metals.
Standard Anodizing Process Flow
A systematic anodizing procedure maintains a consistent rate of oxide growth and an excellent surface. It starts with pre-treatment, which involves cleaning of the metal with alkaline or solvent-based solutions to get rid of oils, dirt, and machining residues. This is etched, giving a thin surface layer, and then desmuted to give a uniform matte finish, and finally anodized to remove any residual alloying elements that can disrupt the anodization process.
The basic anodizing process is done by placing the part in an electrolyte bath and adding direct current. Aluminum atoms react with oxygen ions that are released at the anode to produce aluminum oxide. Current density, temperature, and time are some of the process parameters that control the thickness and structure of this oxide layer.
The porous oxide layer can be dyed, after anodizing, in case of coloration being needed. Sealing is the last step and it uses hot water or steam, which moisturizes the layer of oxide and seals its pores. This process is important in making it corrosion-resistant and fixing any color applied.
The Five Main Types of Anodizing Processes
Anodowanie kwasem siarkowym
The most popular type of aluminum anodizing is sulfuric acid anodizing. It gives rise to a rather thin porous oxide surface, which is perfect for dyeing and ornamental processes. It is common in consumer electronics, architectural elements, and in general industrial components. Its benefits are that it is cost-effective and versatile, but it has moderate wear resistance to harder coatings.
Hard Anodizing
Type III anodizing (also referred to as hard anodizing) utilizes reduced temperatures and increased current densities. This forms a coarse, high-density oxide-coated layer that is very hard and wear-resistant [2]. It is typically used in aerospace parts, auto parts, and high-wear tooling. Nonetheless, it is also costlier and may lead to darker and lighter finishes.
Anodowanie kwasem chromowym
Chromic acid anodizing forms a thinner oxide overlay having better corrosion resistance and reduced dimensional distortion. It is popular in aerospace applications where fatigue resistance is critical. It is not as appropriate to use in the decorative context and is associated with environmental issues because of the presence of hexavalent chromium.
Oxalic Acid Anodizing
Oxalic acid anodizing is a less popular process that offers a more corrosion-resistant and harder surface than sulfuric acid anodizing. In some cases, it is employed in industrial applications where increased durability is needed. The procedure is more complicated and less popular.
Special Anodizing for Titanium and Magnesium
Titanium anodizing is quite different from aluminum anodization. Rather than forming a thick coating of protection, it forms thin layers of oxides which produce interference colors without dyes. This finds extensive application in medical implants and decorative applications. Magnesium anodizing is concerned with enhancing the corrosion resistance because magnesium is very reactive and easily degrades.
Key Process Parameters Affecting Quality
A close relationship between chemical, electrical, and metallurgical variables defines the performance and consistency of Anodizing. These parameters need fine controls in industrial anodizing of aluminum, especially in high-specification work like aerospace parts, molding tools, and electronic enclosures.
Electrolyte Composition
One of the factors that has the greatest impact on the process of anodizing is the electrolyte composition. When anodizing with sulfuric acid, manufacturers use a concentration of 150-220 g/L, although higher concentrations of the acid can be used, which will result in thinner and more porous coats. Lower concentrations will be more likely to give thicker coats, though they may be less uniform. Organic acids or wetting agents stabilize the process and enhance surface finish. Control of contamination is also paramount; the amount of dissolved aluminum in the bath should be checked since too much can decrease the quality of the coating and result in defects like burning or pitting.
Material Composition and Alloy Effects
The composition of materials adds another dimension of complexity. Alloying elements give different aluminum alloys different reactions to anodization [3]. An example is that, in general, alloys in the 6000 series (Al-Mg-Si) form homogeneous and attractive oxide coats, which can be anodized in a decorative manner. Conversely, 2000 series alloys (Al-Cu) might produce darker, less corrosion-resistant finishes because of copper content, and high-silicon casting alloys might produce uneven coloration and low coating integrity. The final anodized surface is also affected by grain structure, previous heat treatment, and machined level finish.
Agitation and Solution Circulation
Agitation and circulation of solution in the electrolyte bath can be neglected, and are necessary to ensure there is an even distribution of ions and temperature. The agitation may be poor, causing localized heat gradients and thus non-uniform coating thickness or defects. Equally, good fixturing and electrical contact provide a uniform distribution of current across the workpiece, especially when the shape of the parts or cross-sectional variations are complex.
Anodizing Time and Process Optimization
Lastly, time is a controlling factor that interacts with others. The longer the anodizing periods, the thicker the oxide becomes, but beyond a given limit, the dissolution and growth are balanced, hindering further growth. Over time, this may also result in lower quality of coating, either through over-etching or widening of the pores. Process optimization, therefore, entails a trade-off between time and present density, temperature, and electrolyte chemistry to obtain the desired characteristics of the coating.
What are the Advantages of Aluminum Anodizing?
Aluminum anodizing provides a set of mechanical, chemical, and aesthetic properties that have seen it being used as a favorite surface treatment in a variety of industries. The development of a coating that is part of the base material is one of its main strengths. The anodic oxide does not peel or delaminate as do paints or electroplated layers, and thus, it is much more durable in harsh environments.
Mechanically, anodized layers (particularly those formed by hard anodizing) offer significant advantages in surface hardness and wear resistance. This is especially useful in tooling components like molds and jigs, where repeated contact and friction would otherwise cause degradation of untreated aluminum [4]. The oxide layer is also highly resistant to corrosion, especially when well sealed, and hence it finds use in the outdoor and marine world.
Another powerful benefit is functionality. Secondary treatment of the porous structure of the anodic layer can be done by dyeing, sealing, or uneven impregnation with lubricants. It allows electrical insulation, thermal management, or decorative finishes to be customized. In electronics, anodized aluminum has the advantage of being both electrically insulating and a conductor of heat, a property that is hard to exhibit with most other coating types.
Anodized materials are aesthetically pleasing, have surface hardness and wear resistance.
What Are the Disadvantages of Aluminum Anodizing?
There are certain disadvantages to anodizing. Its limitation to certain metals is one of its inherent limitations. Although anodization of aluminum is an efficient process, it is not possible to apply the process to steels, copper alloys, or most other engineering metals. It restricts its use in assemblies of mixed materials.
The coatings are also rather brittle in comparison with the base metal, though hard. The layer of oxide may crack in case of high impact or severe deformation, which may undermine the aesthetic and protective quality. It renders anodized surfaces unsuitable in situations with high mechanical shock or flexing.
Limiting factors can also be process complexity and cost. It is necessary to control numerous parameters accurately (such as electrolyte chemistry, temperature, and electrical input) to obtain consistent and high-quality results. The hard anodizing, especially, requires specialized equipment and cooling systems that consume a lot of energy, raising the cost of operation.
Depending on the nature of the anodizing process applied, there might be environmental and regulatory issues. Chromic acid anodizing is an example of a process that utilizes hazardous chemicals that need to be handled, disposed of, and followed up on strictly. Waste treatment and water consumption in the sulfuric acid systems need to be handled well to ensure that the environmental requirements are achieved.
Quality Inspection Standards and Methods
Quality inspection Anodizing is a very important process that guarantees the end anodized coating to be both functional and attractive. Since anodizing may find application in high-performance devices like aerospace parts, high-precision tooling, and electronic enclosures, inspection should be rigorous and standardized.
Coating Thickness Measurement
One of the main quality measures is the coating thickness, measured with non-destructive eddy current tools. The devices are common in production settings as they are fast and precise. To further examine this, cross-sectional microscopy can be used, which involves mounting a fragment of the anodized component, polishing it, and examining it under a microscope to directly measure the thickness of the layer of oxide and assess its structure. Depending on use, the thickness is required, and decorative anodizing typically is between 5-25 microns, and hard anodizing is between 25-100 microns or greater.
Surface Hardness Testing
Another important parameter is surface hardness, particularly in wear-resistant applications. The mechanical strength of the anodized layer is determined by microhardness tests, either Vickers or Knoop hardness tests. Such tests give an idea about the resistance of the coating to abrasion and deformation, which is especially valuable in cases of components with repeated mechanical contacts.
Seal Quality Assessment
The quality of the seal is an important issue that has a direct impact on the resistance to corrosion and durability. The effectiveness of sealing is assessed in a number of ways. Dye stain testing, in which a dye is placed on the anodized surface, will result in any incomplete pore closure being visible as the dye being absorbed into the surface. The other common procedure is the admittance test, which is used to test the properties of the oxidized layer to assess how effectively the sealing of the pores has been done. It also conducts boiling water immersion tests where the component is subjected to hot water and then examined in terms of the surface qualities.
Adhesion Verification
Adhesion testing is not as important as in the coating-based process, but it can be done to verify the integrity of the oxide layer. Because the anodized layer is part of the base material, adhesion tests are not frequently needed, but mechanical means, like scratch testing, can be employed in quality assurance, particularly of critical parts.
Kontrola wyglądu
The appearance inspection on the surface is a significant element in the use of aesthetics. The visual inspection can be performed under controlled lighting conditions to determine the uniformity of colors, surface finish, degree of gloss, and occurrence of defects like streaks or burns. In colored anodizing, spectrophotometers can be employed to quantitatively determine consistency of color in production batches and ensure that the processed batches adhere to strict design specifications.
Process Control and Industry Standards
Modern quality systems involve process control, documentation, and traceability. Electrolyte composition, temperature records, current density plots, and processing time per batch are recorded in production records. The statistical process control techniques commonly used to check the variability and provide uniform output. International standards like ISO 7599 on decorative anodizing and ISO 10074 on hard anodizing include specifications in detail regarding coating properties, testing, and acceptance criteria.
Common Defects and Their Remedies
Defects may also occur during the anodizing due to inappropriate cleaning, uneven distribution of the current, or inappropriate choice of alloy [5]. The local increased temperatures and rough surfaces of burning can be alleviated by decreasing the current density or enhancing agitation. Inequity in coloration may be caused by poor surface preparation or by alloy non-uniformity, and these problems may be overcome by improved pre-treatment control.
Contamination of the electrolyte or improper sealing may result in pitting and corrosion defects. Frequent bath upkeep and adequate sealing procedures are important remedial measures. Streaking and patchiness are associated with inconsistent electrical contact or bad racking design.
Anodizing vs. Other Surface Treatments
Anodizing is contrasted with electroplating, spray coating, and nitriding. It is superior to electroplating in that the substrate is modified, rather than a separate layer of metal deposited, leading to increased adhesion and longevity. Spray coating is flexible about material selection, but is not as hard or wear-resistant as anodized surfaces.
The process of nitriding, which is normally applied to steels, improves the surface hardness by diffusion of nitrogen, though not with aluminum. Anodizing technique is used when dealing with aluminum parts that need to be corrosion-resistant, have moderate wear resistance, and aesthetic enhancements. It is not suitable when there is a need for extreme hardness, thick coating, or when it has to be applied to non-reactive metals.
The choice of anodizing in industrial practice is based on the compatibility of materials with the performance requirements and cost. In the case of aluminum-based systems, anodization of aluminum is still one of the most effective and universal surface engineering solutions that can be adopted.
Referencje
[1] Velling, A. (2021, March 02). Aluminium Anodising. https://fractory.com/aluminium-anodising/
[2] Protolabs (2026). What is anodizing and how does it work? https://www.hubs.com/knowledge-base/what-is-anodizing/
[3] O’Neill, B. (2025, December). The Anodizing Process Explained. https://www.wevolver.com/article/the-anodizing-process-explained
[4] Geomiq (2026). Aluminium Anodising Process and Benefits. https://geomiq.com/blog/aluminium-anodising-process-and-benefits/
[5] HLH Rapid (2025). The Anodizing Process Explained. https://hlhrapid.com/knowledge/the-anodizing-process-explained/